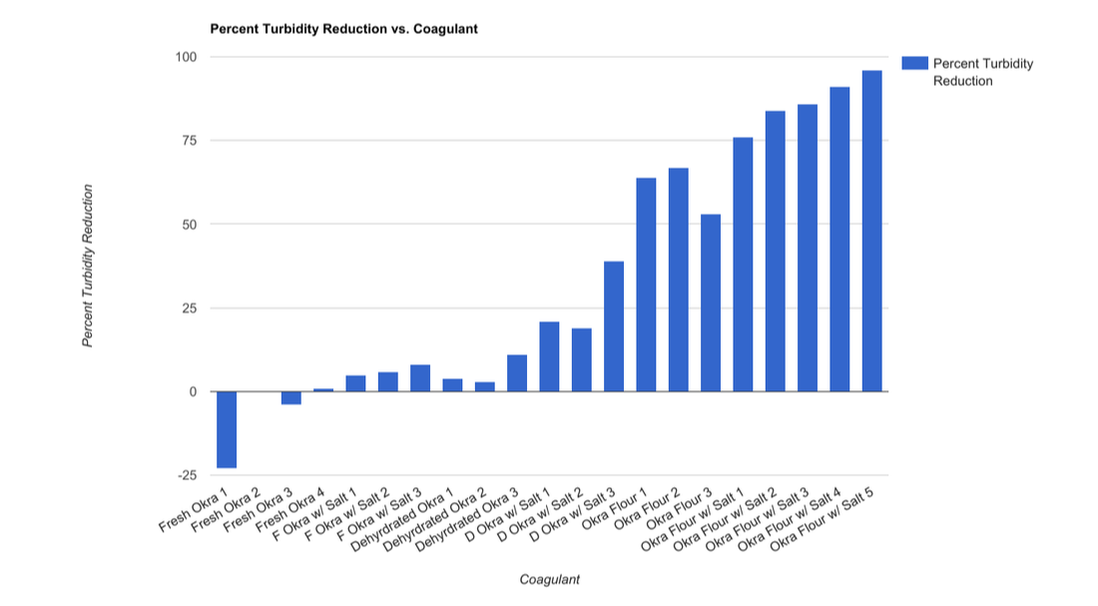
Above is a compilation of our data into a simplified percent turbidity reduction. To make the chart above, we calculated the percent turbidity for all 7 jars in each jar test and graphed the best jar from each test. The formula used to determine percent turbidity reduction was (Final Turbidity/ Initial Turbidity)*100.
Fresh Okra:
The fresh okra test series were started with coagulants made from fresh okra. All 3 tests used fresh okra blended and added to water. The solution was mixed and then added in varying amounts to the jars where a coagulation mixing model was run to determine effectiveness of coagulating polymers in the solution. The first test used 100 mL of fresh okra in 1 mL of water. Then various amounts of coagulant was added to each jar test. This solution actually caused an increase in turbidity due to the amount of raw okra added to the water. Some coagulation was observed; however, the flocks could not be removed from the solution because they remained suspended due to the viscous nature of the coagulant prototype. The second test used 50 mL of fresh okra and 1 L of water, the third used 40 mL of fresh okra, and the fourth 30 mL of fresh okra. Then various amounts of coagulant was added to each jar test. All four also resulted in a viscous agent that did not yield an effective coagulative process.
Fresh Okra with Salt:
After doing research, we determined that adding salt to the coagulant will help draw out the polymers into our agent and will ionize the particulate to encourage effective coagulation. The fresh okra with salt test series were a replicate of the above process with 20 g of salt added to each coagulant. Then various amounts of coagulant was added to each jar test. We tried to get the thinner regions of the coagulant into the syringe instead of the more viscous regions due to the mess that resulted from the mucilage in the previous series. More coagulation was observed in these tests; however, the flocks still remained suspended from getting caught in the mucilage.
Dehydrated Okra:
We thought that perhaps dehydrating the okra would allow us to draw out the polymers without also adding the mucilage to our testing. For the dehydrated okra test series, the fresh okra was chopped and then dehydrated and ground up. It would be prudent to note that the dehydrated okra was incredibly difficult to grind up. Then for each coagulant test, various amounts of dehydrated okra (20g, 50g, and 60 g) was added to one liter of water. Then various amounts of coagulant was added to each jar test. In each of the tests, coagulation was observed; however, only a very small amount. By no means was clean water yielded.
Dehydrated Okra with Salt:
For the dehydrated okra with salt test series, we used the final test from the dehydrated okra series as a preliminary model. Each coagulant consisted of 60g of dehydrated okra, 1 liter of water, and varying amounts of salt(20g, 40g, and 60g). Then various amounts of coagulant was added to each jar in the jar test. After the stirring series was completed, comparatively significant coagulation and flocks were observed; especially in the 60g of salt test. Despite this, clean water was still not yielded.
Okra Flour:
After reading further FTIR Spectroscopy scans ran at the University of Nigeria showed a concentration of the glycoside, protein, and ester polymer concentration in the seeds of the okra so we decided to continue our testing using the seeds. This was convenient because the seeds lack the mucilage and structuring that was messy and got in the way of coagulation. The okra seeds were ground into a "flour" and added in varying amounts(20g,40g, and 60g) to 1 liter of water. Then various amounts of coagulant was added to each jar test. Extremely more impressive coagulation was seen in these tests and significantly cleaner water was produced when compared to the initial condition of the water; however, we knew that theoretically, when salt is added to the coagulant, more polymers will be drawn out so that more coagulation would result. Due to this, we continues with our next series of prototypes: okra flour with salt.
Okra Flour with Salt:
In this testing series, we first had to determine our ratios for the coagulant and then optimize the coagulation without wasting coagulant via fine tuned testing. For the first test, we added 30g of okra flour and 15g of salt to 1 liter of water and then various amounts of coagulant was added to each jar test. For the second test we added 20g of okra flour and 40g of salt to 1 liter of water and then various amounts of coagulant was added to each jar test. We were happy with this coagulant so from there we just had to determine the ratio of coagulant to dirty water that optimized coagulation without waste. The next three tests consisted of fine tuning the coagulant added. The final result was that 14 mL of coagulant added per liter of water would result in 96% turbidity reduction. The practical translation of this prototype is that 20g of okra flour and 40g of salt is equivalent to about 1/8 cup of each added to 1 liter of water.
Fresh Okra:
The fresh okra test series were started with coagulants made from fresh okra. All 3 tests used fresh okra blended and added to water. The solution was mixed and then added in varying amounts to the jars where a coagulation mixing model was run to determine effectiveness of coagulating polymers in the solution. The first test used 100 mL of fresh okra in 1 mL of water. Then various amounts of coagulant was added to each jar test. This solution actually caused an increase in turbidity due to the amount of raw okra added to the water. Some coagulation was observed; however, the flocks could not be removed from the solution because they remained suspended due to the viscous nature of the coagulant prototype. The second test used 50 mL of fresh okra and 1 L of water, the third used 40 mL of fresh okra, and the fourth 30 mL of fresh okra. Then various amounts of coagulant was added to each jar test. All four also resulted in a viscous agent that did not yield an effective coagulative process.
Fresh Okra with Salt:
After doing research, we determined that adding salt to the coagulant will help draw out the polymers into our agent and will ionize the particulate to encourage effective coagulation. The fresh okra with salt test series were a replicate of the above process with 20 g of salt added to each coagulant. Then various amounts of coagulant was added to each jar test. We tried to get the thinner regions of the coagulant into the syringe instead of the more viscous regions due to the mess that resulted from the mucilage in the previous series. More coagulation was observed in these tests; however, the flocks still remained suspended from getting caught in the mucilage.
Dehydrated Okra:
We thought that perhaps dehydrating the okra would allow us to draw out the polymers without also adding the mucilage to our testing. For the dehydrated okra test series, the fresh okra was chopped and then dehydrated and ground up. It would be prudent to note that the dehydrated okra was incredibly difficult to grind up. Then for each coagulant test, various amounts of dehydrated okra (20g, 50g, and 60 g) was added to one liter of water. Then various amounts of coagulant was added to each jar test. In each of the tests, coagulation was observed; however, only a very small amount. By no means was clean water yielded.
Dehydrated Okra with Salt:
For the dehydrated okra with salt test series, we used the final test from the dehydrated okra series as a preliminary model. Each coagulant consisted of 60g of dehydrated okra, 1 liter of water, and varying amounts of salt(20g, 40g, and 60g). Then various amounts of coagulant was added to each jar in the jar test. After the stirring series was completed, comparatively significant coagulation and flocks were observed; especially in the 60g of salt test. Despite this, clean water was still not yielded.
Okra Flour:
After reading further FTIR Spectroscopy scans ran at the University of Nigeria showed a concentration of the glycoside, protein, and ester polymer concentration in the seeds of the okra so we decided to continue our testing using the seeds. This was convenient because the seeds lack the mucilage and structuring that was messy and got in the way of coagulation. The okra seeds were ground into a "flour" and added in varying amounts(20g,40g, and 60g) to 1 liter of water. Then various amounts of coagulant was added to each jar test. Extremely more impressive coagulation was seen in these tests and significantly cleaner water was produced when compared to the initial condition of the water; however, we knew that theoretically, when salt is added to the coagulant, more polymers will be drawn out so that more coagulation would result. Due to this, we continues with our next series of prototypes: okra flour with salt.
Okra Flour with Salt:
In this testing series, we first had to determine our ratios for the coagulant and then optimize the coagulation without wasting coagulant via fine tuned testing. For the first test, we added 30g of okra flour and 15g of salt to 1 liter of water and then various amounts of coagulant was added to each jar test. For the second test we added 20g of okra flour and 40g of salt to 1 liter of water and then various amounts of coagulant was added to each jar test. We were happy with this coagulant so from there we just had to determine the ratio of coagulant to dirty water that optimized coagulation without waste. The next three tests consisted of fine tuning the coagulant added. The final result was that 14 mL of coagulant added per liter of water would result in 96% turbidity reduction. The practical translation of this prototype is that 20g of okra flour and 40g of salt is equivalent to about 1/8 cup of each added to 1 liter of water.